Search Results
FDA has granted “emergency-use authorization” to Eli Lilly’s coronavirus antibody treatment
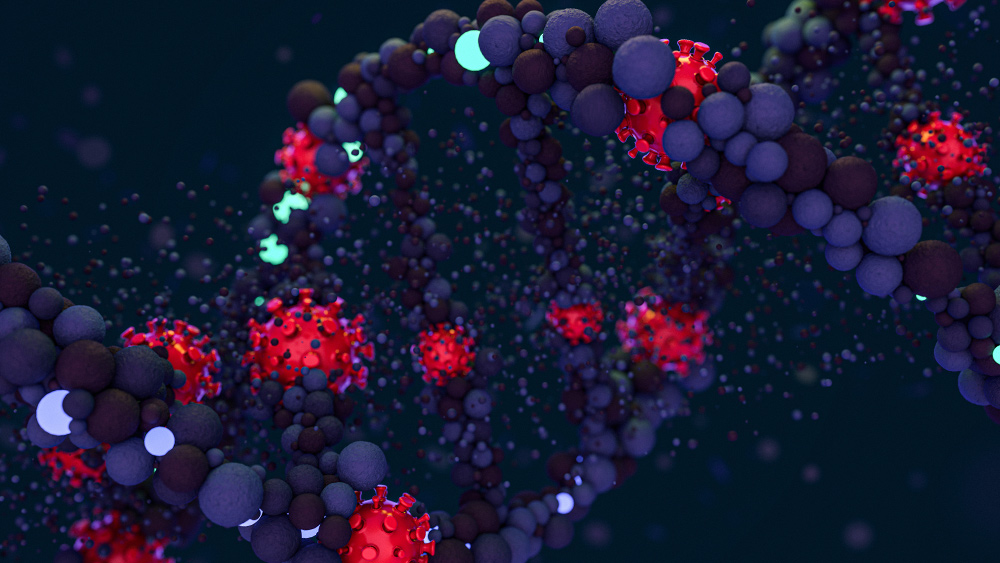 https://www.naturalnews.com/2020-11-12-fda-eli-lilly-coronavirus-antibody-drug.html
November 12, 2020 - The FDA extended the emergency approval to the experimental therapy known as “bamlanivimab.” It may be given to people 12 years old and older who have mild to moderate cases of the coronavirus and have not visited the emergency room or have been admitted to a hospital. Bamlanivimab is a one-time treatment that is administered through an IV.In a statement, FDA Commissioner Stephen M. Hahn gave his support for the approval of bamlanivimab.Bamlanivimab is known ...
https://www.naturalnews.com/2020-11-12-fda-eli-lilly-coronavirus-antibody-drug.html
November 12, 2020 - The FDA extended the emergency approval to the experimental therapy known as “bamlanivimab.” It may be given to people 12 years old and older who have mild to moderate cases of the coronavirus and have not visited the emergency room or have been admitted to a hospital. Bamlanivimab is a one-time treatment that is administered through an IV.In a statement, FDA Commissioner Stephen M. Hahn gave his support for the approval of bamlanivimab.Bamlanivimab is known ...
Medical scientists cautiously describe 100% CURE for cancer using new monoclonal antibody treatment
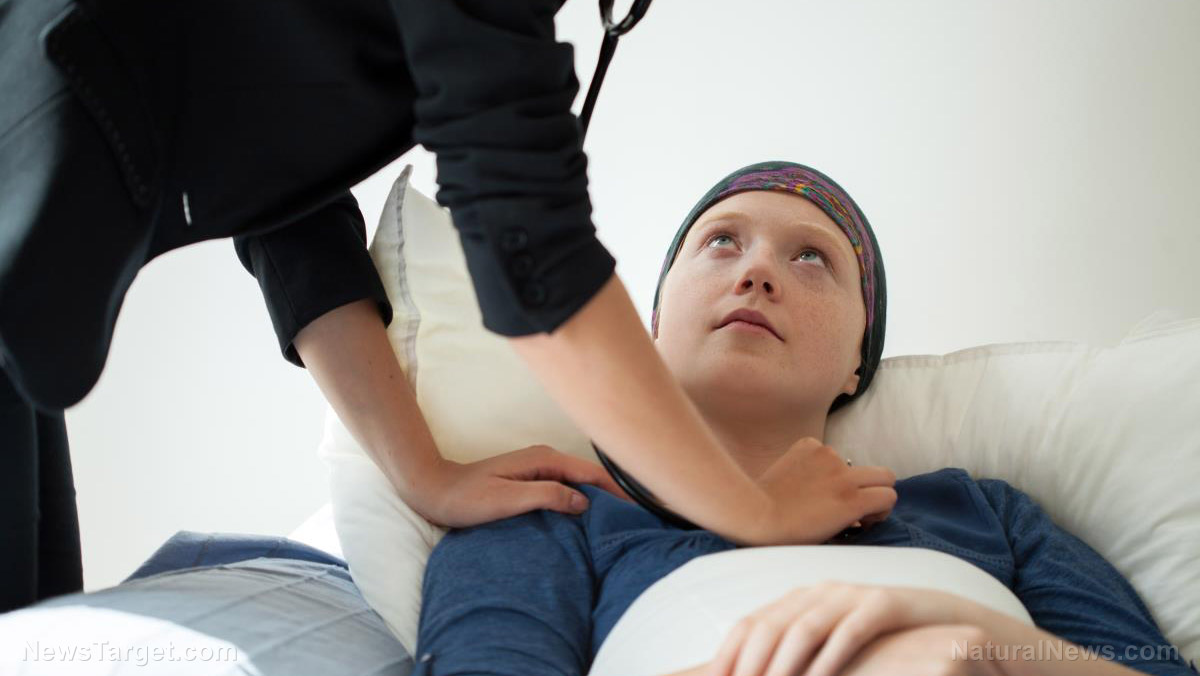 https://www.naturalnews.com/2022-06-09-scientists-cure-cancer-new-monoclonal-antibody-treatment.html
June 09, 2022 - The results of the small trial are already being hailed as "remarkable" and "unprecedented" because all 18 participants are now cancer-free, thanks to receiving dostarlimab every three weeks for six months.One of the patients is now two years out of the trial and still cancer-free, which Dr. Luis A. Diaz Jr., the study's author, told The New York Times "is the first time this has happened in the history of cancer."Fellow co-author Dr. Andrea Cercek, an oncologist ...
https://www.naturalnews.com/2022-06-09-scientists-cure-cancer-new-monoclonal-antibody-treatment.html
June 09, 2022 - The results of the small trial are already being hailed as "remarkable" and "unprecedented" because all 18 participants are now cancer-free, thanks to receiving dostarlimab every three weeks for six months.One of the patients is now two years out of the trial and still cancer-free, which Dr. Luis A. Diaz Jr., the study's author, told The New York Times "is the first time this has happened in the history of cancer."Fellow co-author Dr. Andrea Cercek, an oncologist ...
Drug maker Eli Lilly strikes $375 million deal with US government for COVID-19 antibody treatment
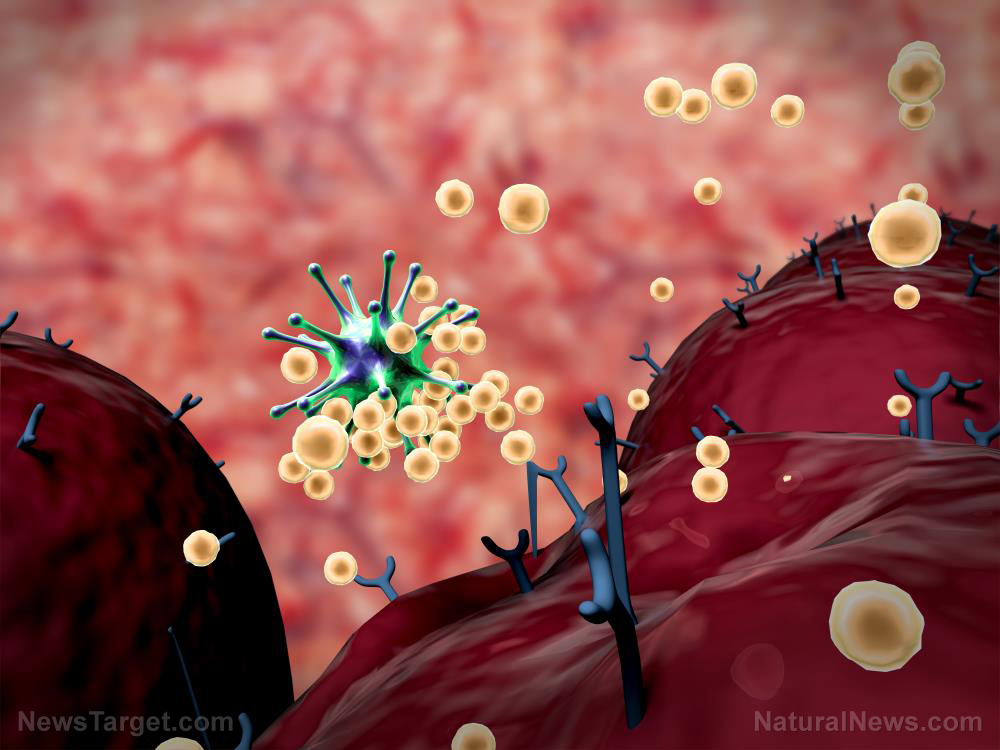 https://www.naturalnews.com/2020-11-02-eli-lilly-strikes-deal-covid-antibody-treatment.html
November 02, 2020 - antibody treatments are showing promise in the treatment of COVID-19Antibody drugs are designed to imitate or enhance the body's immune response to fight infection, compared to vaccines that train the body's immune system to recognize and combat pathogens. Antibody treatments against COVID-19 such as bamlanivimab produce a temporary effect that could help prevent worsening symptoms in infected patients.The company reported in September that bamlanivimab ...
https://www.naturalnews.com/2020-11-02-eli-lilly-strikes-deal-covid-antibody-treatment.html
November 02, 2020 - antibody treatments are showing promise in the treatment of COVID-19Antibody drugs are designed to imitate or enhance the body's immune response to fight infection, compared to vaccines that train the body's immune system to recognize and combat pathogens. Antibody treatments against COVID-19 such as bamlanivimab produce a temporary effect that could help prevent worsening symptoms in infected patients.The company reported in September that bamlanivimab ...
FDA identifies dozens of inaccurate coronavirus antibody tests
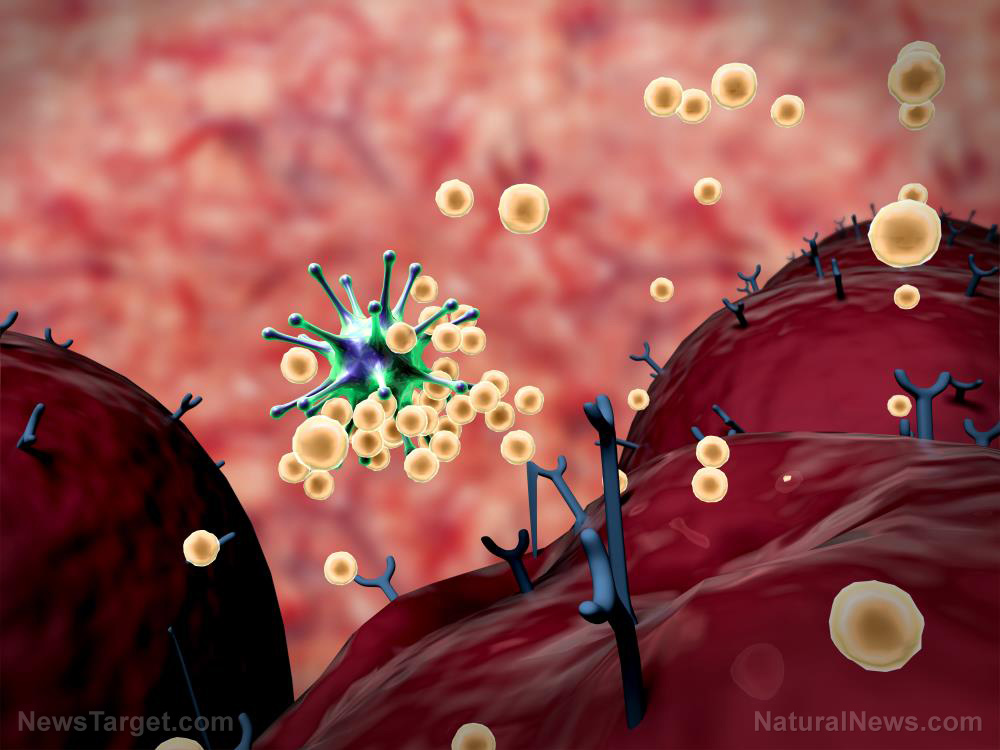 https://www.naturalnews.com/2020-06-01-fda-identifies-dozens-of-inaccurate-coronavirus-antibody-tests.html
June 01, 2020 - A total of 27 vendors will no longer be allowed to sell their antibody tests because they haven’t been shown to work. Yesterday, the FDA published a list of the makers whose tests will no longer be marketed or distributed; it’s not known whether they will be facing additional penalties. Many of them appear to be from Chinese companies.In tests carried out by ABC News and the Mayo Clinic, half of the antibody tests examined were found to be unreliable, with one even saying ...
https://www.naturalnews.com/2020-06-01-fda-identifies-dozens-of-inaccurate-coronavirus-antibody-tests.html
June 01, 2020 - A total of 27 vendors will no longer be allowed to sell their antibody tests because they haven’t been shown to work. Yesterday, the FDA published a list of the makers whose tests will no longer be marketed or distributed; it’s not known whether they will be facing additional penalties. Many of them appear to be from Chinese companies.In tests carried out by ABC News and the Mayo Clinic, half of the antibody tests examined were found to be unreliable, with one even saying ...
AstraZeneca’s experimental “antibody cocktail” fails to prevent coronavirus symptoms
 https://www.naturalnews.com/2021-06-21-astrazeneca-antibody-cocktail-fails-prevent-symptoms-coronavirus.html
June 21, 2021 - The trial, named "Storm Chaser," was on Phase III. It involved 1,121 adult volunteers from the United States and the United Kingdom. The vast majority of the volunteers did not have COVID-19 symptoms at the start of the trial. But they were exposed to the coronavirus soon after the trial started.AstraZeneca claimed the goal of the trial was to figure out whether the long-acting antibody cocktail it developed could protect people who had recently been in contact with coronavirus-positive ...
https://www.naturalnews.com/2021-06-21-astrazeneca-antibody-cocktail-fails-prevent-symptoms-coronavirus.html
June 21, 2021 - The trial, named "Storm Chaser," was on Phase III. It involved 1,121 adult volunteers from the United States and the United Kingdom. The vast majority of the volunteers did not have COVID-19 symptoms at the start of the trial. But they were exposed to the coronavirus soon after the trial started.AstraZeneca claimed the goal of the trial was to figure out whether the long-acting antibody cocktail it developed could protect people who had recently been in contact with coronavirus-positive ...
AstraZeneca begins advanced trials for COVID-19 antibody medication
 https://www.naturalnews.com/2020-10-26-astrazeneca-begins-advanced-trials-covid-antibody-medication.html
October 26, 2020 - antibody treatmentsAstraZeneca received financial support of about $486 million from the U.S. government to aid its antibody-drug development; in turn, the company will produce an initial 100,000 doses of AZD7742 by the end of 2020. A separate agreement permitted the federal government to acquire up to one million doses in 2021.The $486 million agreement follows an earlier $25 million deal between the company, the Defense Advanced Research Projects Agency and ...
https://www.naturalnews.com/2020-10-26-astrazeneca-begins-advanced-trials-covid-antibody-medication.html
October 26, 2020 - antibody treatmentsAstraZeneca received financial support of about $486 million from the U.S. government to aid its antibody-drug development; in turn, the company will produce an initial 100,000 doses of AZD7742 by the end of 2020. A separate agreement permitted the federal government to acquire up to one million doses in 2021.The $486 million agreement follows an earlier $25 million deal between the company, the Defense Advanced Research Projects Agency and ...
FDA panel unanimously supports new RSV antibody drug for infants
 https://www.naturalnews.com/2023-06-14-fda-panel-supports-rsv-antibody-drug-infants.html
June 14, 2023 - All 21 members of the FDA's Antimicrobial Drugs Advisory Committee (AMDAC) unanimously voted in favor of the monoclonal antibody nirsevimab on June 8. The committee agreed that the benefits of nirsevimab exceed the dangers in preventing RSV infections for newborns and infants in their initial RSV season. With the AMDAC's decision, nirsevimab is closer to receiving final regulatory approval.In a separate 19-2 vote, the AMDAC supported the monoclonal antibody's use in children ...
https://www.naturalnews.com/2023-06-14-fda-panel-supports-rsv-antibody-drug-infants.html
June 14, 2023 - All 21 members of the FDA's Antimicrobial Drugs Advisory Committee (AMDAC) unanimously voted in favor of the monoclonal antibody nirsevimab on June 8. The committee agreed that the benefits of nirsevimab exceed the dangers in preventing RSV infections for newborns and infants in their initial RSV season. With the AMDAC's decision, nirsevimab is closer to receiving final regulatory approval.In a separate 19-2 vote, the AMDAC supported the monoclonal antibody's use in children ...
FDA criticized for restricting the use of COVID antibody treatments
 https://www.naturalnews.com/2022-01-27-fda-criticized-for-restricting-covid-antibody-treatments.html
January 27, 2022 - antibody treatments. While the FDA noted that the drugs are currently not cleared for use in any of the U.S. states or territories, they may be authorized in other regions if proven to work against new variants.Doctors also have alternate therapies to battle COVID-19 cases, including new antiviral pills from Merck and Pfizer. However, they are currently still in short supply. Another antibody drug from GlaxoSmithKline that is effective against COVID is also in ...
https://www.naturalnews.com/2022-01-27-fda-criticized-for-restricting-covid-antibody-treatments.html
January 27, 2022 - antibody treatments. While the FDA noted that the drugs are currently not cleared for use in any of the U.S. states or territories, they may be authorized in other regions if proven to work against new variants.Doctors also have alternate therapies to battle COVID-19 cases, including new antiviral pills from Merck and Pfizer. However, they are currently still in short supply. Another antibody drug from GlaxoSmithKline that is effective against COVID is also in ...
Eli Lilly monoclonal antibody drug trial paused over safety concerns
 https://www.naturalnews.com/2020-10-20-eli-lilly-drug-trial-paused-safety-concerns.html
October 20, 2020 - antibody treatments being pushed, but is it too soon?The company is one of several pursuing experimental treatments for COVID-19 that use “monoclonal antibodies.” These are mass-produced copies of the antibodies that the immune system produces in response to the coronavirus. (Related: Possible coronavirus treatment may come from antibodies produced by… llamas?)Antibodies can block the coronavirus from infecting healthy cells and preliminary data from testing ...
https://www.naturalnews.com/2020-10-20-eli-lilly-drug-trial-paused-safety-concerns.html
October 20, 2020 - antibody treatments being pushed, but is it too soon?The company is one of several pursuing experimental treatments for COVID-19 that use “monoclonal antibodies.” These are mass-produced copies of the antibodies that the immune system produces in response to the coronavirus. (Related: Possible coronavirus treatment may come from antibodies produced by… llamas?)Antibodies can block the coronavirus from infecting healthy cells and preliminary data from testing ...
This Ayurvedic herb can regulate immune function and stimulate antibody production
 https://www.naturalnews.com/2021-04-20-ayurvedic-herb-regulates-immune-function-stimulate-antibody-production.html
April 20, 2021 - Besides skin conditions, herbal medicines containing black cutch extracts are used to treat a wide variety of diseases, including asthma, bronchitis, colic, diarrhea, ulcers, anemia and stomatitis (mouth inflammation). Recent studies have also found that the bark, heartwood and sap of this useful plant have many pharmaceutical properties, such as anthelmintic, antihyperglycemic, antipyretic and anti-inflammatory activities.In a recent study, researchers at Mahatma Gandhi ...
https://www.naturalnews.com/2021-04-20-ayurvedic-herb-regulates-immune-function-stimulate-antibody-production.html
April 20, 2021 - Besides skin conditions, herbal medicines containing black cutch extracts are used to treat a wide variety of diseases, including asthma, bronchitis, colic, diarrhea, ulcers, anemia and stomatitis (mouth inflammation). Recent studies have also found that the bark, heartwood and sap of this useful plant have many pharmaceutical properties, such as anthelmintic, antihyperglycemic, antipyretic and anti-inflammatory activities.In a recent study, researchers at Mahatma Gandhi ...
Doubts raised on effectiveness of coronavirus antibody testing in the UK
 https://www.naturalnews.com/2020-07-15-doubts-on-coronavirus-antibody-testing-uk.html
July 15, 2020 - According to a review of studies conducted by Cochrane, a U.K.-based network that assesses health research to help direct policy, roughly one in 10 antibody tests will miss cases of COVID-19. This could lead to people making harmful decisions delaying treatment or even traveling.“There is no decision [patients] should be making based on the results of a test,” stated the review’s lead author Jon Deeks, a professor of biostatistics at Birmingham University in England. “We ...
https://www.naturalnews.com/2020-07-15-doubts-on-coronavirus-antibody-testing-uk.html
July 15, 2020 - According to a review of studies conducted by Cochrane, a U.K.-based network that assesses health research to help direct policy, roughly one in 10 antibody tests will miss cases of COVID-19. This could lead to people making harmful decisions delaying treatment or even traveling.“There is no decision [patients] should be making based on the results of a test,” stated the review’s lead author Jon Deeks, a professor of biostatistics at Birmingham University in England. “We ...
Covid “negative” patients are overwhelming hospitals, exactly as predicted with Antibody Dependent Enhancement
 https://www.naturalnews.com/2021-11-10-covid-negative-patients-overwhelming-hospitals-ade.html
November 10, 2021 - According to the talking-heads, these people do not have the Wuhan coronavirus (Covid-19) because many of them are "fully vaccinated," so the whole thing is a "mystery," they claim.Since the jabs are "safe and effective," there is no explanation for this latest crisis. It must just be a coincidence, right?"ERs are now swamped with seriously ill patients – but many don't even have covid," reported Michigan Radio.NPR also relayed the story, pretending ...
https://www.naturalnews.com/2021-11-10-covid-negative-patients-overwhelming-hospitals-ade.html
November 10, 2021 - According to the talking-heads, these people do not have the Wuhan coronavirus (Covid-19) because many of them are "fully vaccinated," so the whole thing is a "mystery," they claim.Since the jabs are "safe and effective," there is no explanation for this latest crisis. It must just be a coincidence, right?"ERs are now swamped with seriously ill patients – but many don't even have covid," reported Michigan Radio.NPR also relayed the story, pretending ...
alternative treatment
https://www.naturalnews.com/tag/alternative-treatment.html a long time ago - Natural.NewsNatural Health News & Self-Reliance Doctors look to alternative treatments like medicinal cannabis as World Cancer Day approaches World Cancer Day is rapidly approaching as more and more scientists seek natural alternatives that can fight cancer, without damaging healthy cells. One alternative that has received considerable attention in the last year is medicinal cannabis. It’s been known for years that marijuana can help ease the symptoms of the disease, but ...natural treatment
https://www.naturalnews.com/tag/natural-treatment.html a long time ago - Natural.NewsNatural Health News & Self-Reliance Melatonin, the sleep hormone, may treat MS Melatonin is known by many for its ability to help you relax and go to sleep. A new study shows that melatonin may also be beneficial for people with multiple sclerosis (MS) – a debilitating neurological disorder. The root cause of MS is unknown, but we do know that it causes the body to attack its own neurons Read More Top 10 medicinal plants for natural sunburn relief Medicinal ...Moderna to develop booster shot after South African strain decreased its coronavirus vaccine’s antibody levels
 https://www.naturalnews.com/2021-02-10-moderna-to-develop-coronavirus-vaccine-booster-shot.html
February 10, 2021 - Laboratory analysis showed that the Moderna jab had six times less antibodies that block the virus from human cell receptors when tested against the South African B1351 strain. That amount of antibodies can still provide protection, albeit on a shorter period. Moderna is now working on a booster shot for the South African strain, which is going into pre-clinical trials as of writing.The pharmaceutical firm said its vaccine worked just as well against the B117 variant ...
https://www.naturalnews.com/2021-02-10-moderna-to-develop-coronavirus-vaccine-booster-shot.html
February 10, 2021 - Laboratory analysis showed that the Moderna jab had six times less antibodies that block the virus from human cell receptors when tested against the South African B1351 strain. That amount of antibodies can still provide protection, albeit on a shorter period. Moderna is now working on a booster shot for the South African strain, which is going into pre-clinical trials as of writing.The pharmaceutical firm said its vaccine worked just as well against the B117 variant ...
cancer treatment
https://www.naturalnews.com/tag/cancer-treatment.html a long time ago - All posts tagged with cancer treatmentThe spicy, active ingredient in chilies found to kill off cancer cellsTestosterone cures cancer; doctors stunned at discoveryCould venom from an Irish spider be the cure to many diseases?New Chemo-free treatment uses body’s own immune system to attack cancer cells Natural News Toolbar Privacy Policy Terms of Use About Us Contact Us/Feedback Write for Natural News Media ...Mental Treatment (comic)
https://www.naturalnews.com/021430.html a long time ago - Mental Treatment (comic)Comments by Mike Adams, the Health RangerFor example, IG Farben, which committed numerous crimes against humanity and was charged with enslavement, torture and extermination of prisoners, was dissolved by the Nuremberg War Tribunal and split into three companies:Carl Wurster served as chairman of the board for BASF through 1974. During WWII, he was on the board of the company that manufactured Zyklon-B gas.Helmut Kohl, the ...Chemotherapy "Treatment" (comic)
https://www.naturalnews.com/026330_cancer_child.html a long time ago - Chemotherapy "Treatment" (comic)Comments by Mike Adams, the Health RangerBilly's recovery proves cancer doctors wrongWho are the real fugitives from the law?Never take your children to a conventional cancer doctorRelated CounterThink Cartoons:Related Articles:Take Action: Support NaturalNews.comAdvertise with NaturalNews...Vilcabamba Ecuador resourcesFree Special ReportsSupport NaturalNews Sponsors:Advertise with ...Mental Treatment (comic)
https://www.naturalnews.com/021430_modern_psychiatry.html a long time ago - Mental Treatment (comic)Comments by Mike Adams, the Health RangerFor example, IG Farben, which committed numerous crimes against humanity and was charged with enslavement, torture and extermination of prisoners, was dissolved by the Nuremberg War Tribunal and split into three companies:Carl Wurster served as chairman of the board for BASF through 1974. During WWII, he was on the board of the company that manufactured Zyklon-B gas.Helmut Kohl, the ...Chemotherapy "Treatment" (comic)
https://www.naturalnews.com/026330.html a long time ago - Chemotherapy "Treatment" (comic)Comments by Mike Adams, the Health RangerBilly's recovery proves cancer doctors wrongWho are the real fugitives from the law?Never take your children to a conventional cancer doctorRelated CounterThink Cartoons:Related Articles:Take Action: Support NaturalNews.comAdvertise with NaturalNews...Vilcabamba Ecuador resourcesFree Special ReportsSupport NaturalNews Sponsors:Advertise with ...Free Email Alerts
Get independent news alerts on natural cures, food lab tests, cannabis medicine, science, robotics, drones, privacy and more.
About Us
Who We Are
Meet the Health Ranger
Free Email Newsletter
Support
Write for Us
Media Information
Advertising Information
Submit a News Tip
Privacy Policy
Terms of Use
Links
Home
All News
Health Ranger Store
Health Ranger Report Podcast
Natural News App
BrightAnswers.ai
Blog Articles
RSS Feed
Follow Us
This site is part of the Natural News Network © All Rights Reserved. Privacy | Terms All content posted on this site is commentary or opinion and is protected under Free Speech. Truth Publishing International, LTD. is not responsible for content written by contributing authors. The information on this site is provided for educational and entertainment purposes only. It is not intended as a substitute for professional advice of any kind. Truth Publishing assumes no responsibility for the use or misuse of this material. Your use of this website indicates your agreement to these terms and those published here. All trademarks, registered trademarks and servicemarks mentioned on this site are the property of their respective owners.



