According to the CDC's own data, around 42 percent of Americans between the ages of five and 17 have already been infected with and recovered from the coronavirus. This means that American youth are probably already at herd immunity without the need for vaccinations.
Even the World Health Organization (WHO) understands that COVID-19 cases among under 18-year-olds only represent a sliver of all reported cases. And among those cases, most experience a very mild form of the disease and even fewer die from it.
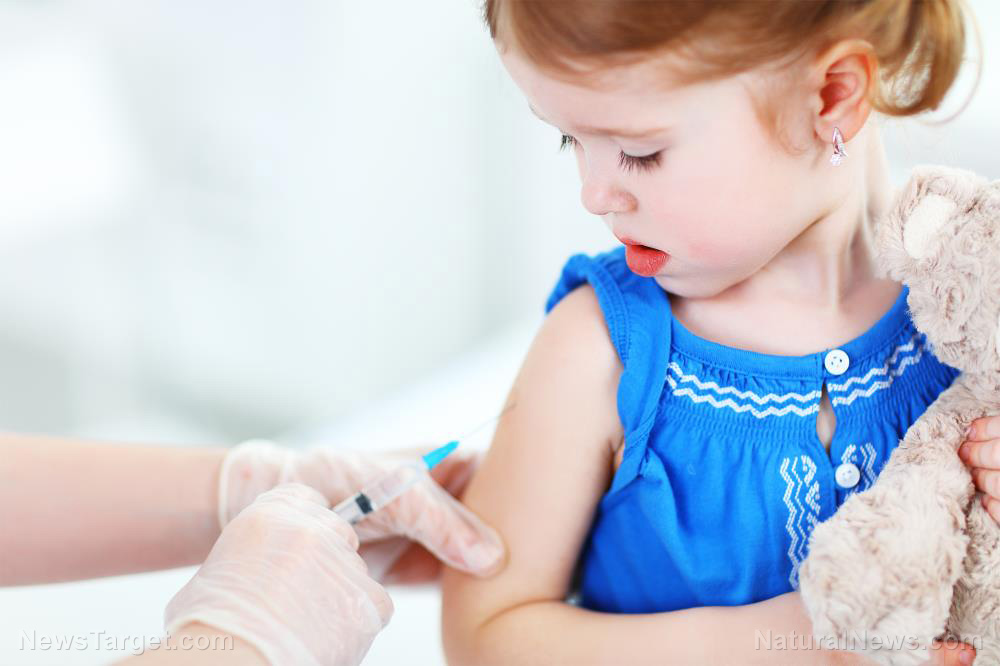
But companies like Pfizer are still pushing forward with their vaccine trials for children, with the approval of the CDC. Some hospitals are even expanding the trial to include "volunteers" as young as six months old.
"Now that the vaccine has proven to be effective in adults, it is a natural next step in the research process to study the unique needs of younger individuals," said Dr. Julia Garcia, director of clinical infectious diseases research at the Ochsner Medical Center in Louisiana. (Related: Pfizer pushes ahead with plans to push coronavirus vaccines on younger children, including toddlers and infants.)
"A child's immune system is different than an adult's immune system, so it is critically important to have a study focused on the efficacy of the vaccine in this cohort," she added.
One couple from Louisiana signed up all their children for Pfizer's trial at Ochsner. The parents are Drs. Cuong Bui and Erin Biro and they signed up their three children. The eldest is six, the middle child is three and the youngest is just 14 months old.
Biro claimed she was doing this so that her kids can live "safely in a world where we don't have to worry about them getting sick from COVID."
"We're super excited that our entire family now has the opportunity and the chance of possibly being protected … and also just contributing to the research and the science to hopefully get all kids across the finish line," she added.
WHO believes children should not be vaccinated against COVID-19 right now
On Tuesday, June 22, the WHO issued new guidance recommending that parents hold off on vaccinating their children against the coronavirus for the time being.
The WHO said children are less likely to get the virus. Even if they do, they will only most likely experience a very mild form of it compared to adults and are not in urgent need of vaccinations unless they have pre-existing conditions.
Because of this, the WHO believes vaccines should be prioritized for those with health conditions, healthcare and other frontline workers and older individuals.
"More evidence is needed on the use of the different COVID-19 vaccines in children to be able to make general recommendations on vaccinating children against COVID-19," wrote the WHO in its guidance.
Despite this guidance, the WHO still believes in the supposed effectiveness of vaccines. The WHO's Strategic Advisory Group of Experts said that the Pfizer-BioNTech coronavirus vaccine is suitable for children aged 12 years old and above.
It also did not recommend that the ongoing Pfizer COVID-19 vaccine trials on children should be stopped. The organization is instead waiting for the results of the trial and will most likely support the vaccination of children once the trial's results come out.
Instead of getting the COVID-19 vaccine, the WHO is recommending parents continue giving their children other childhood vaccines.
Sources include:
Please contact us for more information.