According to a poll, public trust in Oxford-AstraZeneca’s coronavirus vaccine dipped across Europe after leaders delayed the rollout of the vaccine due to fears that it causes blood clots in recipients. It wasn't clear if the poll had been conducted before or after the European Medicines Agency (EMA) ruled that the Oxford-AstraZeneca vaccine wasn't linked to a higher clotting risk.
YouGov poll shows Europeans are cautious about Oxford-AstraZeneca vaccine
In a separate YouGov study conducted back in February, results revealed that Europeans were already cautious about the Oxford-AstraZeneca vaccine. Poll takers were more confident in the alternatives like the Pfizer and Moderna vaccines, although more people in general considered the Oxford-AstraZeneca vaccine to be safe than unsafe in all countries studied except for France.
But in a more recent YouGov poll, results showed that more people are worried that the Oxford-AstraZeneca vaccine is unsafe. Figures increased to 61 percent in France, 55 percent in Germany and 43 percent in Italy. The percentage of worried poll takers in France increased by 18 points since February. In Italy, the increase was even greater at 27 points.
In Spain, only 38 percent of people considered the Oxford-AstraZeneca vaccine safe for use. (Related: Italian prosecutors open MANSLAUGHTER investigation as man dies a day after getting AstraZeneca’s coronavirus vaccine.)
Despite the news about the vaccine causing blood clots, confidence in the vaccine only fell by nine percent among poll takers in the U.K. Over three-quarters of Britons still think both the AstraZeneca and Pfizer vaccines are safe.
Poll results showed that in Sweden, the AstraZeneca vaccine is still considered by more people as safe than not (43 percent and 34 percent, respectively). In Denmark, those who considered Oxford-AstraZeneca safe and unsafe for use are tied at 42 percent. However, more people consider Pfizer and Moderna safe compared to Oxford-AstraZeneca in both Sweden and Denmark.
Some European countries also suggested that the Oxford-AstraZeneca vaccine should only be administered to people under 65 due to a lack of trial data.
Matt Smith, YouGov's lead data journalist, explained that the European leaders’ concerns over the vaccine may have damaged its reputation among the poll takers.
Women more likely to develop side effects from Oxford-AstraZeneca vaccine
In one study conducted by Lareb, a Dutch side effects research organization, researchers analyzed reports from nearly 4,000 women aged 21 to 65 who were given a coronavirus vaccine. Results revealed women are far more likely to develop side effects after receiving the Oxford-AstraZeneca vaccine compared to Pfizer-BioNTech's Comirnaty.
In the study, researchers examined the voluntary reports submitted by 3,331 female participants who were given a first dose of the AstraZeneca vaccine and 599 who received the first dose of the Comirnaty vaccine.
About two-thirds of women who received the AstraZeneca vaccines reported symptoms like fatigue, headaches, muscle strain and generally feeling unwell. At least 39 percent of the volunteers reported that they developed a fever of over 38 C, which lasted for up to two days. The figure was almost twice the percentage of women who reported symptoms like fatigue, headaches, or muscle strain and triple the rate of women who said they felt sick.
Meanwhile, only five percent of women given the Comirnaty vaccine reported having a fever for a short duration. More than 3,600 of the female participants are healthcare workers.
Oxford-AstraZeneca issues guidance related to its vaccine's "very rare" side effect
AstraZeneca has issued guidance to patients who may experience a "very rare" side effect from its coronavirus vaccine.
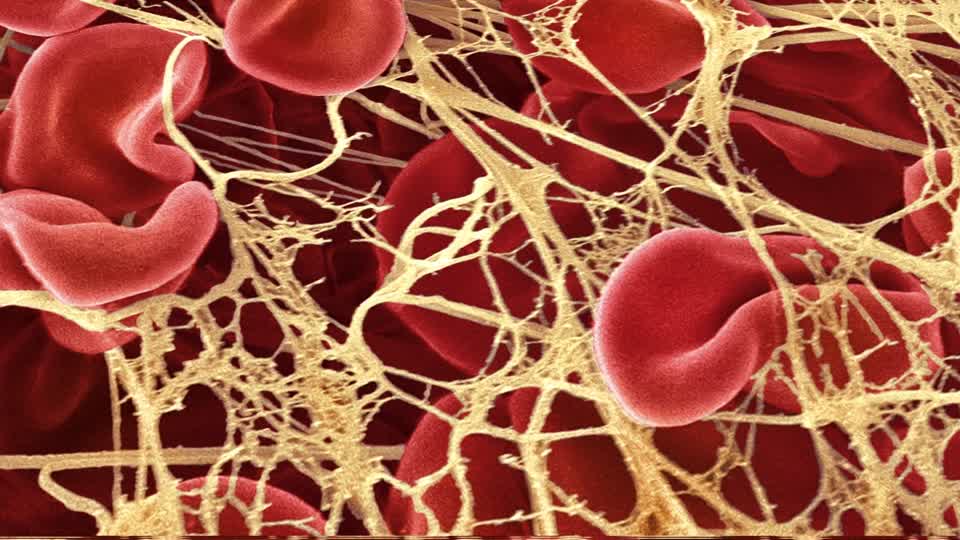
In a letter sent by the company to Irish healthcare professionals, AstraZeneca gave advice on what to do if patients experience symptoms associated with conditions like thrombosis and thrombocytopenia.
The guidance follows several reports of blood clotting in patients who received the company's vaccine in European countries despite the vaccine being ruled as safe as investigations by the EMA.
In agreement with the EMA and the Health Products Regulatory Authority (HPRA), AstraZeneca formulated guidance for Irish patients as a precaution. According to the letter from the company, the benefits of the vaccine outweigh the "very rare" risk of blood clots with low blood platelets after a patient has been inoculated.
AstraZeneca also advised healthcare professionals and patients to be alert to the symptoms of thromboembolism and thrombocytopenia. Vaccinated patients were urged to seek immediate medical attention if they experience the following symptoms after vaccination:
- Chest pain
- Leg swelling
- Persistent abdominal pain
- Shortness of breath
Patients with neurological symptoms like severe or persistent headaches and blurred vision after vaccination, or those who experience skin bruising beyond the site of vaccination after several days were also advised to seek "prompt medical attention."
Healthcare professionals were instructed to report any suspected adverse reactions after the use of the vaccine "in accordance with national reporting guidelines."
In Ireland and other countries across Europe, the use of the vaccine was paused for several days earlier in March after the Norwegian Medicines Agency announced severe side effects in four patients. Officials in Ireland reported that the decision to pause the use of the Oxford-AstraZeneca vaccine was made on a precautionary principle.
Despite these reported cases of adverse effects, both the EMA and the National Public Health Emergency Team (NPHET) urged that the use of the vaccine should continue, with the EMA saying that the vaccine is "safe and effective."
Professor Karina Butler of Ireland's National Immunization Advisory Committee (NIAC) said the reports of thrombosis were small in number and were considered "very rare adverse events."
Visit VaccineInjuryNews.com for detailed reports on the negative side effects of coronavirus vaccines.
Sources include:
Please contact us for more information.