Leaked documents show Merck knew of Vioxx dangers, yet hid them for years
Saturday, August 06, 2005 by: Dani Veracity
Tags: Merck, Vioxx, bad medicine
- Newly released JFK files reveal Pentagon's role in creating Lyme disease and covid in the same lab
- Dr. Suzanne Humphries makes bombshell appearance on Joe Rogan podcast, exposing vaccine industry deception back to POLIOMYELITIS
- L.A.'s rebuilding nightmare: Only 4 permits issued after fire destroys 6,000 homes
- Discovery of vast underground city beneath Giza pyramids challenges human history
- Black cumin seed oil emerges as a powerful ally against breast cancer and chronic inflammation
- PROCESSED TABLE SALT in foods found to fuel depression
- Ginseng's hidden anti-aging power: How compound K is rewriting the rules of skincare
- Here are TEN all-natural ways to protect your garden without using harmful chemicals
- “Independent” anti-Russia outlet MEDUZA faces COLLAPSE as US funding dries up
- The hidden battle in your glass: How A1 and A2 milk could shape your health
- Catastrophic 7.7 earthquake devastates Myanmar and Thailand; death toll could reach 100,000
- BPA: The hidden hormone disruptor sabotaging your health - and how to fight back
- CDC finally halts $11 billion COVID funding scam as health officials admit the ‘pandemic’ was a fraud
- AI breakthrough slashes celiac disease diagnosis time from months to minutes
- Europe braces for WAR as EU urges citizens to STOCKPILE FOOD, in latest provocations with Russia
- “Endgame: The Hidden Agenda 21” unveils a world of conspiracy and control
- Putin vows to 'finish off' Ukraine, accuses West of prolonging the war
- Former Congresswoman exposes CCP's deep infiltration of California through universities, ports, and fentanyl
- Newly released JFK files reveal Pentagon's role in creating Lyme disease and covid in the same lab
- Analysis: The coming economic collapse, a mass uprising and Trump's three secret weapons to halt the growing revolt
- Festive flavors: The sweet history, nutritional profile and health benefits of pecan pie
- Elon Musk: Aliens could be here on Earth RIGHT NOW
- Trump reverses course on Gaza plan, says “nobody is expelling Palestinians”
- Big Pharma's $8 Billion bribery scheme exposed: how doctors are pushed to prescribe junk science, not heal
- Boys are back in town: Trump’s patriotic alpha crew takes the wheel while toxic females ride in the backseat
- Reclaim your health: How midlife exercise reverses years of inactivity
- A lack of integrity in Academia: Harvard professor found GUILTY of fraudulent research to promote CRT theory
- Survival 101: Effective EMF blocking techniques
- EPA advisor admits the agency is funneling billions to climate groups ahead of Trump’s return to White House
- Dr. Mike Yeadon releases 15-minute testimony - WATCH - about genocidal intent of COVID “vaccines”
- 5 Simple steps to boost your brainpower: How to strengthen executive function in a distracted world
- Florida takes a stand: DeSantis proposes permanent ban on mRNA vaccine mandates
- Sugarcane extract superior to cholesterol-lowering drugs?
- Mike Adams Sermon 66: God will DESTROY ISRAEL for its wickedness
- Pilots report mysterious lights 'moving at extreme speeds' across Oregon skies
- Space war brewing? Russia threatens to destroy Starlink satellites
- EPA advisor admits the agency is funneling billions to climate groups ahead of Trump’s return to White House
- California's social media censorship law struck down: A victory for free speech or a threat to online safety?
- The Health Ranger releases “Vaccine Zombie” song and music video, using AI-animated zombies for the music video
- Dr. Mike Yeadon releases 15-minute testimony - WATCH - about genocidal intent of COVID “vaccines”
- The pandemic as a tool for INDOCTRINATION: Understanding “The Indoctrinated Brain” by Dr. Michael Nehls
- Newly released JFK files reveal Pentagon's role in creating Lyme disease and covid in the same lab
- Florida takes a stand: DeSantis proposes permanent ban on mRNA vaccine mandates
- Mike Adams releases country western hit single: Goin’ Back in Time is Comin’ Home
- Mike Adams releases music poetry sensation: A Child of God
- “Why we influenced the 2020 elections”: Facebook files reveal the coordinated effort to bury the Hunter Biden laptop story
- Unpacking the Lies That We’ve Been Fed – new song and music video released by Mike Adams, the Health Ranger
- RFK Jr. clears key hurdle: Sen. Susan Collins backs controversial HHS nominee, signaling a new era for health policy
- Mike Adams releases new song and music video: Nothing More Disgusting Than a Globalist
- Michigan sheriff announces criminal investigation into 2020 election crimes, Dominion Voting Systems
- Israeli soldiers accused of even more torture and abuse in the West Bank
- Migrants are taking advantage of recent hurricanes to scam residents and loot their homes
- House Intelligence Committee calls for the ARREST and PROSECUTION of Dr. Anthony Fauci
- Rep. Nancy Mace introduces bill to ban biological males from female facilities on federal property
- Red Cross issues warning to stop blood plasma donations from vaccinated people
- Scientists confirm: GENIUS brain function can be spontaneously unleashed in humans without any apparent cause
- EPA advisor admits the agency is funneling billions to climate groups ahead of Trump’s return to White House
- HYSSOP: What research reveals about the health benefits of this ancient holy herb
- Two containers with completed ballots fall out of truck in Florida
- Fully vaccinated about to see “tsunami” of illness and death, warns virologist
- Global leaders unite to clamp down on “misinformation” with UN-backed Cascais Declaration
- BREAKING: 2025 NDAA authorizes mandatory military draft of WOMEN across America… as Pentagon pursues global NUCLEAR war with both Russia and China at the same time
- Michael Yon warns of a ZIONIST TAKEOVER in Trump’s second administration
- BOMBSHELL: DNA testing kits are a SCAM to develop ethnic-specific bioweapons
- Ozempic and Wegovy weight loss drugs are injectable LIZARD VENOM PEPTIDES that may unleash a devastating wave of organ failure… side effects align with symptoms of SNAKE BITES
- Israeli soldiers accused of even more torture and abuse in the West Bank
- These 13 countries just signed an agreement to engineer a global FAMINE by destroying food supply
- NASA admits that climate change occurs because of changes in Earth’s solar orbit, and NOT because of SUVs and fossil fuels
- RFK Jr. clears key hurdle: Sen. Susan Collins backs controversial HHS nominee, signaling a new era for health policy
- Sermon 30: How Jesus reveals Caesar’s FAKE CURRENCY and FALSE AUTHORITY
- Coriander seeds: Ancient medicine backed by modern science
- Arizona officials claim Maricopa County needs 10-13 days to tabulate results of the election
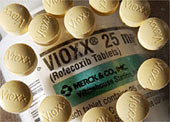
An application for a patent seems harmless enough, except when the product in question is a correcting reformulation of the now-withdrawn prescription drug Vioxx. In this leaked document, the authors specifically stated that Vioxx increases the risk of potentially fatal cardiovascular disease by reducing the body's production of a substance called prostacyclin, which prevents platelet coagulation. This reduction may alter the ratio of prostacyclin to thromboxane, a substance which can constrict blood vessels, resulting in excessive blood clotting, and, consequently, heart attacks and strokes -- the very disorders that Merck denies Vioxx promotes.
In other words, based on this evidence, Merck has apparently been feigning ignorance of Vioxx's disastrous complications for the last five years. Their knowledge of the risk wasn't cursory, either. In fact, as early as March 30, 2000, Edward Scolnick, the former head of Merck's research labs, realized the risk was high enough to suggest that Vioxx be combined with an anti-clotting agent. Then, in 2001, Merck filed an application with the U.S. Patent Office for a new-and-improved Vioxx that was to be combined with a thromboxane inhibitor, but Merck eventually dropped the project and the patent. In response to the leaked document concerning the proposed patent, Merck's law firm released a statement saying that the application had nothing to do with Merck's firm belief that Vioxx was safe; however, the statement seems illogical: Why would Merck wish to fix what wasn't broken?
How to bury scientific truth
On Nov. 1, 2004, the eastern edition of The Wall Street Journal broke the story: "Warning Signs: Emails Suggest Merck Knew Vioxx's Dangers at Early Stage," discrediting Merck's feigned ignorance of Vioxx's cardiovascular risks. According to a memo dated Nov. 21, 1996, Merck officials first "wrestled" with the issue of Vioxx's dangerous effects in 1996 when they considered running a trial to demonstrate that Vioxx is gentler on the digestive system than other painkillers, like aspirin. Officials feared that the study would also reveal Vioxx's cardiovascular risk because the subjects of course would not be able to avoid the risk by taking aspirin. The controversy continued into 1997, when Merck official Briggs Morrison sent an email dated Feb. 25, 1997, arguing that, unless test subjects received aspirin, the revealed cardiovascular risks would "kill [the] drug."Now, let's examine Merck's concerns for a moment: Based on leaked ducuments, Merck officials knew that Vioxx posed cardiovascular risk as early as 1996 and, yes, they were worried about it, because if it were revealed, the risk might decrease sales. In response to Morrison's email, Alise Reicin, who is now Merck's vice president for clinical research, emailed that the company was in a "no-win situation" and proposed that people with high risk of cardiovascular problems be excluded from the story, so that the difference between the rate of cardiovascular problems associated with Vioxx and other drugs "would not be evident."
(In other words, it's not okay for people with increased cardiovascular risk to take Vioxx in a research study that will have publicized results, but it seemed perfectly alright for them to take Vioxx by prescription and suffer or die in private.)
Then, on March 9, 2000, a year after the FDA's approval of Vioxx, Scolnick sent his colleagues an email in which he said that Vioxx's cardiovascular side effects "are clearly there" and are a "shame." However, four years later, Gilmartin still had the gall to say that the research findings that finally initiated Vioxx's ban were "unexpected." This apparent lie could have gone unnoticed, if it hadn't been for The Wall Street Journal's breaking story. It was noticed, however, and it has sparked a series of media coverage and stock market losses. "Can the news get any uglier for Merck?" asked the November 6, 2004 issue of Economist. Well, thanks to the latest leaked document, it just did.
Merck is going to have a difficult time backing out of this corner. In the New Jersey Vioxx lawsuit where the unscrupulous communication by Merck was first revealed, the judge called the document privileged information, so it was not used. Other judges might not consider it privileged, thus allowing it in their courtrooms. According to Associated Press reporter Theresa Agovino, analysts estimate that Merck's liability could reach as high as $18 billion. Based on current evidence, Merck ignored research findings in order to make more money; now, they may lose more than they ever imagined.
Sources on the recent Vioxx leak (in order of appearance in this article):
"The Payoff and the Company that 'Really Puts Patient Safety First'"
One slide was captioned, "The Payoff." "What are the long-term benefits of your being able to bring value to the relationship with our customers?" it asked. Pictured underneath was a sales rep holding a glowing bag of cash captioned "Bonus."
"For Our Eyes Only … Inside Merck's Mess" by Jim Edwards and Michael Applebaum, Brandweek 5/16/2005, page 52
When Merck & Co. pulled its big-selling painkiller Vioxx off the market in September, Chief Executive Raymond Gilmartin said the company was "really putting patient safety first."
"Warning Signs: E-Mails Suggest Merck Knew Vioxx's Dangers at Early Stage; As Heart-Risk Evidence Rose, Officials Played Hardball; Internal Message: 'Dodge!'; Company Says 'Out of Context'" by Anna Wilde Mathews and Barbara Martinez, Wall Street Journal (Eastern edition) 11/1/2004, page A1
"Leaked Communication Reveals Merck Tried to Reformulate Vioxx in 2000 and Patent It in 2001"
Merck & Co. researchers privately sought to reformulate Vioxx in 2000 to reduce its cardiovascular side effects, even as the drug maker was publicly playing down a study that highlighted the pain relief medication's potential heart attack risk, an internal company document shows.
The widely publicized study in March 2000 found that patients taking Vioxx were five times more likely to have heart attacks than individuals using the generic medicine naproxen. Merck insisted at the time that this was a result of naproxen's cardioprotective properties and not any defect in Vioxx.
But behind the scenes, company scientists were considering combining Vioxx with another agent to reduce the risk of heart attacks and strokes, according to a document that was mistakenly provided by Merck to plaintiff lawyers as part of the evidence-gathering process in one of the hundreds of Vioxx lawsuits around the country.
That document, a communication between Merck researchers and the company's patent department, stated that the way Vioxx works to reduce pain might also increase cardiovascular problems. They suggested a patent be sought for a combination drug mixing Vioxx with another agent to lessen the risk.
Merck filed an application with the U.S Patent Office in 2001 to combine Vioxx with what is called a thromboxane inhibitor, according to a statement released Wednesday by the company's counsel, Hughes, Hubbard & Reed. The statement said the hope was that Vioxx could provide cardioprotective protection while preserving its gastrointestinal benefits. But the project was later dropped, making the patent application moot.
"Merck Tried to Alter Vioxx in 2000" by Theresa Agovino, Associated Press
"Researchers Knew that Vioxx May Lead to Cardiovascular Problems"
According to the document, Edward Scolnick, the former head of Merck's research labs, was the first to suggest combining Vioxx with an agent that would block blood platelets from clotting. Such clots can lead to heart attacks and strokes. There are three dates on the document; the first is March 30, 2000.
The document's authors said Vioxx might reduce the production of a substance called prostacyclin, which prevents platelet aggregation. That reduction may alter the ratio of prostacyclin to thromboxane, a substance which can constrict blood vessels and cause clotting. The change may cause increased risk of cardiac and cerebral adverse events, the document said.
"Merck Tried to Alter Vioxx in 2000" by Theresa Agovino, Associated Press
A Nov. 21, 1996, memo by a Merck official shows the company wrestling with this issue. It wanted to conduct a trial to prove Vioxx was gentler on the stomach than older painkillers. But to show the difference most clearly, the Vioxx patients couldn't take any aspirin. In such a trial, "there is a substantial chance that significantly higher rates" of cardiovascular problems would be seen in the Vioxx group, the memo said.
A similar view was expressed in a Feb. 25, 1997, e-mail by a Merck official, Briggs Morrison. He argued that unless patients in the Vioxx group also got aspirin, "you will get more thrombotic events" -- that is, blood clots -- "and kill [the] drug."
In response, Alise Reicin, now a Merck vice president for clinical research, said in an e-mail that the company was in a "no-win situation." Giving study subjects both Vioxx and aspirin, she wrote, could increase the "relative risk," apparently referring to gastrointestinal problems. But, she added, "the possibility of increased CV [cardiovascular] events is of great concern." From the context, it seems Dr. Reicin meant "increased" relative to older drugs.
She added in parentheses: "I just can't wait to be the one to present those results to senior management!" She proposed that people with high risk of cardiovascular problems be kept out of the study so the difference in the rate of cardiovascular problems between the Vioxx patients and the others "would not be evident."
"Warning Signs: E-Mails Suggest Merck Knew Vioxx's Dangers at Early Stage; As Heart-Risk Evidence Rose, Officials Played Hardball; Internal Message: 'Dodge!'; Company Says 'Out of Context'" by Anna Wilde Mathews and Barbara Martinez, Wall Street Journal (Eastern edition) 11/1/2004, page A1
By 2000, one email suggests Merck recognized that Vioxx didn't merely lack the protective features of old painkillers but that something about the drug itself was linked to an increased heart risk. On March 9, 2000, the company's powerful research chief, Edward Scolnick, e-mailed colleagues that the cardiovascular events "are clearly there" and called it a "shame." He compared Vioxx to other drugs with known side effects and wrote, "there is always a hazard." But the company's public statements after Dr. Scolnick's email continued to reject the link between Vioxx and increased intrinsic risk.
"Warning Signs: E-Mails Suggest Merck Knew Vioxx's Dangers at Early Stage; As Heart-Risk Evidence Rose, Officials Played Hardball; Internal Message: 'Dodge!'; Company Says 'Out of Context'" by Anna Wilde Mathews and Barbara Martinez, Wall Street Journal (Eastern edition), 11/1/2004, page A1
"Can the News Get Any Uglier for Merck?"
Can the news get any uglier for Merck? Barely a month after Ray Gilmartin pulled Vioxx, a hitherto blockbuster arthritis drug, from the market, Merck's boss suddenly finds himself fighting for his job. Since he broke the news about Vioxx, citing new evidence that the drug raises the risk of heart attacks and strokes, the firm's share price has nearly halved, knocking over $30 billion off the value of the world's fourth-biggest drug company. This week, plaintiff lawyers leaked to the press an apparently damning e-mail and internal company documents (which were supposedly sealed under court order pending lawsuits involving Vioxx), calling into question Merck's reputation for probity. As the lawsuits pile up, the proud, insular giant is giving every appearance it has no idea what it is dealing with. The firm is "bewildered", says one well-placed source.
"Big trouble for Merck," Economist 11/6/1004, page 61
"Can the Leaked Documents be Used in Court?"
The document became an issue in a New Jersey Vioxx lawsuit when Merck objected to its use on the grounds that it was an attorney-client communication between company scientists and in-house patent counsel.
New Jersey Superior Court Judge Carol E. Higbee ruled May 27 that the document was privileged and could not be used at trial. She also ordered that all copies of the privileged document be returned to Merck or destroyed, but she required Merck to turn over other documents related to the reformulation, including e-mails and memos, within 30 days.
Benjamin Zipursky, a professor at Fordham Law School in New York, said it's conceivable that other judges could rule differently and allow the document to be used in Vioxx cases. Lawyers in the Texas case said they have the document and will attempt to make it part of their evidence.
"Merck Tried to Alter Vioxx in 2000" by Theresa Agovino, Associated Press
Merck at FETCH.news
Get independent news alerts on natural cures, food lab tests, cannabis medicine, science, robotics, drones, privacy and more.
Take Action: Support Natural News by linking to this article from your website
Permalink to this article:
Embed article link: (copy HTML code below):
Reprinting this article:
Non-commercial use OK, cite NaturalNews.com with clickable link.
Follow Natural News on Facebook, Twitter, Google Plus, and Pinterest
Science News & Studies
Medicine News and Information
Food News & Studies
Health News & Studies
Herbs News & Information
Pollution News & Studies
Cancer News & Studies
Climate News & Studies
Survival News & Information
Gear News & Information
News covering technology, stocks, hackers, and more


"Big Tech and mainstream media are constantly trying to silence the independent voices that dare to bring you the truth about toxic food ingredients, dangerous medications and the failed, fraudulent science of the profit-driven medical establishment.
Email is one of the best ways to make sure you stay informed, without the censorship of the tech giants (Google, Apple, Facebook, Twitter, YouTube, etc.). Stay informed and you'll even likely learn information that may help save your own life."
–The Health Ranger, Mike Adams













































