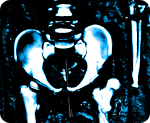
Exposed: Johnson & Johnson hid data showing 40 percent failure rate of hip implants
Thursday, January 31, 2013 by: Ethan A. Huff, staff writer
Tags: Johnson & Johnson, hip implants, failure
- Omega-3s dramatically inhibit breast cancer tumor growth
- Resveratrol may be the missing link to prevent and even treat Alzheimer's disease
- Lymphatic vessels acknowledged in the brain; could be the key to better understanding disease
- How to Detox Fluorides from Your Body
- NaturalNews exclusive: US government developing ultimate cyber weapon; Prime-factoring quantum computing makes encryption obsolete
- A War is against Your Immune System, Part I
- IV ozone Ebola cure documented by Dr. Rowen, then systematically suppressed by government
- Green gardening secrets: How to eliminate bugs and pests without using poison
- Forbes.com writer and biotech shill Jon Entine exposed as violent instigator who physically attacked wife and traumatized daughter - court documents
- Real history: Vaccines not responsible for halting infectious diseases of 20th century
- Bill Gates, Monsanto, and eugenics: How one of the world's wealthiest men is actively promoting a corporate takeover of global agriculture
- How to halt migraines without prescription drugs
- How EMF filters can protect your home
- Artichokes boost brain health, have other incredible health benefits
- Depopulation-advocating globalist Bill Gates comes out in full push for totalitarian socialism, says 'democracy is a problem'
- Unusual tropical fruit proves 10,000 times more effective at treating cancer than chemotherapy
- America: The land of meat, cheese and bread
- Protein found in breast milk may protect infants from deadly gastrointestinal disease
- The REAL FAKE NEWS exposed: '97% of scientists agree on climate change' is an engineered hoax... here's what the media never told you
- The 10 worst toxins hidden in vitamins, supplements and health foods
- Health benefits of taking spirulina daily
- Study: Hot chili pepper compound kills cancer without side effects
- Spirulina: Immune boosting, mood enhancing, protein-rich wonder food
- Beat cancer with 35% hydrogen peroxide
- Omega-3s dramatically inhibit breast cancer tumor growth
- Tetanus vaccines found spiked with sterilization chemical to carry out race-based genocide against Africans
- Three effective natural remedies for varicose veins
- How to heal cavities naturally
- Lymphatic vessels acknowledged in the brain; could be the key to better understanding disease
- Bill Gates, Monsanto, and eugenics: How one of the world's wealthiest men is actively promoting a corporate takeover of global agriculture
- 4 enzyme rich foods that can dramatically improve digestion
- The remarkable health benefits of Epsom salt baths
- Dandelion root extract found to kill leukemia cells, prostate cancer cells and chemo-resistant melanoma
- Bill Gates says vaccines can help reduce world population
- Six different times the despicable leftist media has attacked CHILDREN of Republicans
- Natural ways to move mucus and expel phlegm
- The REAL FAKE NEWS exposed: '97% of scientists agree on climate change' is an engineered hoax... here's what the media never told you
- Red Cabbage Found to Contain 36 Anti-Cancer Anthocyanins
- Beat cancer with 35% hydrogen peroxide
- The 10 worst toxins hidden in vitamins, supplements and health foods
- Forget Filling Cavities: Regrow Your Teeth Instead
- Untested vaccines causing new wave of polio-like paralysis across India
- Bill Gates says vaccines can help reduce world population
- The United Nations 2030 Agenda decoded: It's a blueprint for the global enslavement of humanity under the boot of corporate masters
- Four supplements that will benefit almost everyone
- The best and worst forms of magnesium to take as a supplement
- Canadian agency attacks Jenny McCarthy for demanding mercury-free vaccines
- Apricot Seeds Kill Cancer Cells without Side Effects
- Before his death, father of ADHD admitted it was a fictitious disease
- Vaccines lower immunity
- Top foods, herbs and supplements to keep on hand in the event of a nuclear crisis
- Vitamin E kills off cancer cells and prevents their reproduction, study finds
- 4 enzyme rich foods that can dramatically improve digestion
- New study finds that spirulina can help treat pancreatic cancer
- EPA advisor admits the agency is funneling billions to climate groups ahead of Trump’s return to White House
- Newly released JFK files reveal Pentagon's role in creating Lyme disease and covid in the same lab
- Eleven days before Iran bombed Tel Aviv, my microscope revealed haunting images of EXACTLY what would happen
- Morphic resonance “remote viewing” reveals iconic Middle East images of stealth bombers, a falcon and a one-horned ram
- Mike Adams releases country western hit single: Goin’ Back in Time is Comin’ Home
- DECENTRALIZED SPIRITUALITY and the true teachings of Christ: Overcoming the censorship, threats and lies of organized religion to truly know God and the Universal Christ
- Global leaders unite to clamp down on “misinformation” with UN-backed Cascais Declaration
- Two containers with completed ballots fall out of truck in Florida
- BOMBSHELL: Covid-19 mRNA nanoparticles EMIT LIGHT SIGNALS that communicate MAC addresses used for self-assembly inside the blood vessels
- I Want My Bailout Money – new song released by Mike Adams
- HEALTH SECRETS: How to Instantly Block MSG Toxicity Using Natural Substances (and the secret of Methylene Blue)
- The Health Ranger releases “Vaccine Zombie” song and music video, using AI-animated zombies for the music video
- BOMBSHELL: Internal Pfizer documents exposed and reveal at least 16 PERCENT of their mRNA vaccine "adverse events" are REPRODUCTIVE DISORDERS
- HYSSOP: What research reveals about the health benefits of this ancient holy herb
- BOMBSHELL: DNA testing kits are a SCAM to develop ethnic-specific bioweapons
- Amazing microscopy photos reveal how freezing crystals attempt to mimic electronic structures they are touching
- RFK Jr. clears key hurdle: Sen. Susan Collins backs controversial HHS nominee, signaling a new era for health policy
- The Coming Gold Revaluation: Strategic Financial Realignment in an Era of Dollar Collapse
As reported by The New York Times (NYT), the device in question, known as an Articular Surface Replacement (ASR), was first introduced back in the early 2000s as an alternative to traditional hip implant devices. Its design was supposed to overcome deficiencies inherent in previous models that were made from metal and plastic parts. But a major design flaw in the ASR caused the device's supposedly innovative ball mechanism to grind against the inside cup, which in turn caused it to release metallic debris inside the bodies of many patients who received the implant.
Just a few years after its release by J&J's DePuy Orthopaedics unit, internal tests conducted by company engineers revealed that the device was a complete failure, and that it was unsuitable for continued use. But rather than address the issue and recall the device, J&J continued to market the faulty ASR to surgeons across the country for at least another three years, even as thousands of these same physicians were rejecting the device in favor of safer and more durable alternatives.
"The ASR represents one of the biggest medical device failures in recent decades," writes Barry Meier for NYT about the fiasco. "According to DePuy's internal estimates, it is projected to fail within five years in about 40 percent of patients who received one. That is eight times the failure rate of most orthopedic implants."
J&J eventually issued a recall on the ASR in 2010, but nearly 100,000 patients worldwide had already received the device, and many of these patients are now suing the company for fraud. J&J also failed to admit there was anything wrong with the device during the recall, insisting instead that "poor sales" were responsible for its discontinuation. However, internal records have now revealed that DePuy executives, including company president Andrew Ekdahl, were fully aware of the dangers associated with the ASR and yet did nothing about them.
Patients injured as a result of the faulty implant are being represented by the national law firm Parker Walchman LLP. Both the standard ASR and the resurfacing ASR manufactured by J&J DePuy are included in the lawsuit.
Sources for this article include:
http://www.nytimes.com
http://www.prweb.com
Johnson & Johnson at FETCH.news
Get independent news alerts on natural cures, food lab tests, cannabis medicine, science, robotics, drones, privacy and more.
Take Action: Support Natural News by linking to this article from your website
Permalink to this article:
Embed article link: (copy HTML code below):
Reprinting this article:
Non-commercial use OK, cite NaturalNews.com with clickable link.
Follow Natural News on Facebook, Twitter, Google Plus, and Pinterest
Science News & Studies
Medicine News and Information
Food News & Studies
Health News & Studies
Herbs News & Information
Pollution News & Studies
Cancer News & Studies
Climate News & Studies
Survival News & Information
Gear News & Information
News covering technology, stocks, hackers, and more


"Big Tech and mainstream media are constantly trying to silence the independent voices that dare to bring you the truth about toxic food ingredients, dangerous medications and the failed, fraudulent science of the profit-driven medical establishment.
Email is one of the best ways to make sure you stay informed, without the censorship of the tech giants (Google, Apple, Facebook, Twitter, YouTube, etc.). Stay informed and you'll even likely learn information that may help save your own life."
–The Health Ranger, Mike Adams























